
In the real world there is no nature vs. nurture argument, only an infinitely complex and moment-by-moment interaction between genetic and environmental effects.
-Gabor Mate
Our Research
Our goal is to understand, and provide insight into, the ethanol-sensitive genes that regulate craniofacial development. This insight will lay the groundwork into the mechanistic basis of ethanol teratogenesis.
Why zebrafish? Due to their external fertilization, high fecundity, optical clarity of the embryos and the large variety of genetic tools available, zebrafish is an ideal model to study many developmental disorders. With this tool set, they are ideally suited to screening both genetics and the environment.

Fetal Alcohol Spectrum Disorders

Fetal Alcohol Spectrum Disorders (FASD) describes a wide array of ethanol-induced craniofacial abnormalities, as well as cognitive disabilities.
Many factors contribute to the pathology of FASD, but of these, genetics is the least understood. Our work leverages the high degree of genetic amenability in zebrafish to identify ethanol-sensitive loci involved in craniofacial development.
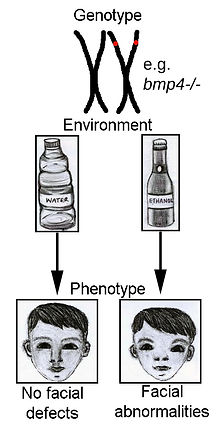
Gene-Ethanol Interactions
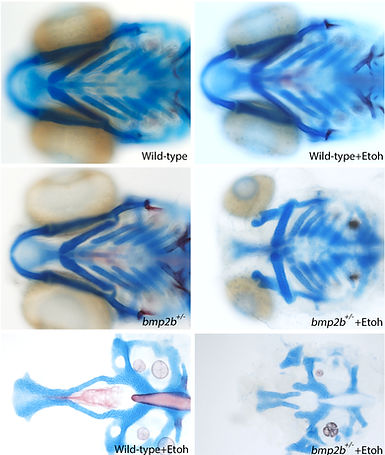
Our approach takes a novel, broad, less biased approach to identify gene-ethanol interactions via genetic screens. From a screen of available zebrafish mutants, we have identified multiple members of the Bone Morphogenetic Protein (Bmp) signaling pathway as being ethanol sensitive exhibiting jaw and palate defects.
Expanding upon our previous screen, we took a unique, unbiased forward genetic approach to identify novel ethanol-sensitive genetic loci. From this screen, we have identified 3 mutants that impact various aspects of craniofacial development.
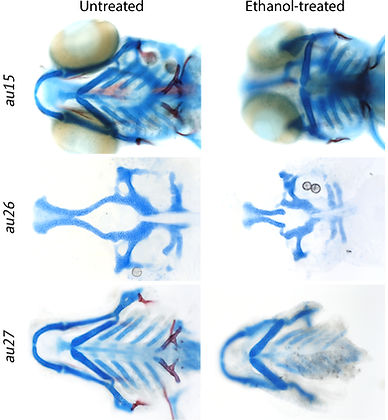
We are currently examining the mechanistic basis of these gene-ethanol interactions and how they impact craniofacial development. We will also continue to identify new ethanol-sensitive genetic loci involved in craniofacial development.
Epithelial Morphogenesis
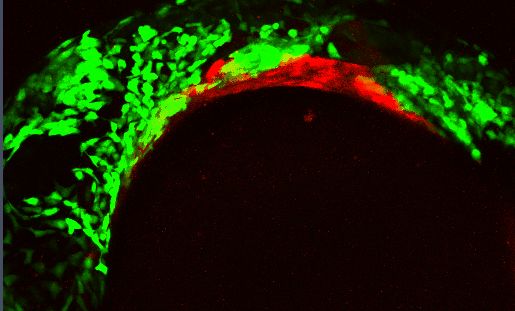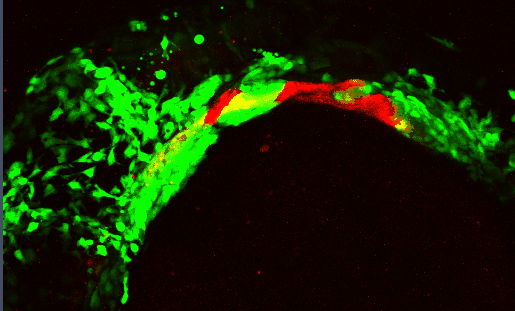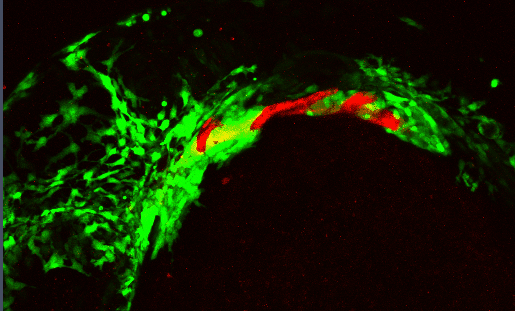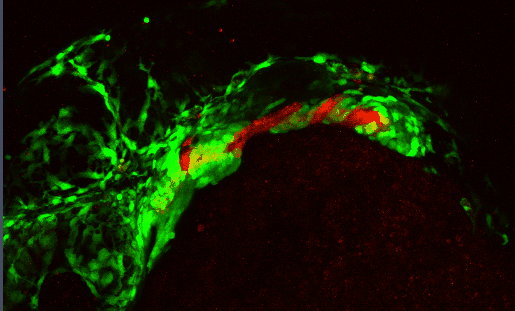
Development of the craniofacial skeleton requires a complex, highly dynamic set of signaling interactions between the cranial neural crest (CNC - which make up the facial skeleton) and the facial epithelia. Proper moprhogenesis of the facial epithelia is critical in establishing these interactions
Several signaling pathways, including the Bone Morphogenetic Protein (Bmp) pathway, regulate these morphogenetic events. Disrupting the Bmp pathway alters these epithelial-CNC interactions perturbing craniofacial development.

We are characterizing the signaling pathways that underlie epithelial morphogenesis and how disruptions to this morphogenetic events impacts development of the craniofacial skeleton.
Publications
-
Variation in phenotypes from a Bmp-Gata3 genetic pathway is modulated by Shh signaling. Swartz ME, Lovely CB, Eberhart JK. PLoS Genet. 2021 May 25;17(5):e1009579. doi: 10.1371/journal.pgen.1009579
-
Animal Models of Gene-Alcohol Interactions. Lovely, CB. Birth Defects Res. 2020 Mar 1;112(4):367-379. doi: 10.1002/bdr2.1623
-
Quantification of Ethanol Levels in Zebrafish Embryos Using Head Space Gas Chromatography. Lovely, CB. J Vis Exp. 2020 Feb 11;(156):10.3791/60766. doi: 10.3791/60766.
-
Novel Ethanol-Sensitive Mutants Identified in an F3 Forward Genetic Screen. Swartz ME, Lovely CB, McCarthy N, Kuka T, Eberhart JK. Alcohol Clin Exp Res Actions 2020 Jan;44(1):56-65. doi: 10.1111/acer.14240
-
Gene-environment interactions in development and disease. Lovely C, Rampersad M, Fernandes Y, Eberhart J. Wiley Interdiscip Rev Dev Biol. 2017 Jan;6(1). doi: 10.1002/wdev.247.
-
Fishing for Fetal Alcohol Spectrum Disorders: Zebrafish as a Model for Ethanol Teratogenesis. Lovely CB, Fernandes Y, Eberhart JK. Zebrafish. 2016 Oct;13(5):391-8. doi: 10.1089/zeb.2016.1270.
-
Bmp signaling mediates endoderm pouch morphogenesis by regulating Fgf signaling in zebrafish. Lovely CB, Swartz ME, McCarthy N, Norrie JL, Eberhart JK. Development. 2016 Jun 1;143(11):2000-11. doi: 10.1242/dev.129379.
-
Pdgfra and Pdgfrb genetically interact during craniofacial development. McCarthy N, Liu JS, Richarte AM, Eskiocak B, Lovely CB, Tallquist MD, Eberhart JK. Dev Dyn. 2016 Jun;245(6):641-52. doi: 10.1002/dvdy.24403.
-
Developmental age strengthens barriers to ethanol accumulation in zebrafish. Lovely CB, Nobles RD, Eberhart JK. Alcohol. 2014 Sep;48(6):595-602. doi: 10.1016/j.alcohol.2014.06.003. Erratum in: Alcohol. 2016 Mar;51:101.
-
Commentary: catching a conserved mechanism of ethanol teratogenicity. Lovely CB, Eberhart JK. Alcohol Clin Exp Res. 2014 Aug;38(8):2160-3. doi: 10.1111/acer.12484.
-
A screen of zebrafish mutants identifies ethanol-sensitive genetic loci. Swartz ME, Wells MB, Griffin M, McCarthy N, Lovely CB, McGurk P, Rozacky J, Eberhart JK. Alcohol Clin Exp Res. 2014 Mar;38(3):694-703. doi: 10.1111/acer.12286.
-
Analyzing craniofacial morphogenesis in zebrafish using 4D confocal microscopy. McGurk PD, Lovely CB, Eberhart JK. J Vis Exp. 2014 Jan 30;(83):e51190. doi: 10.3791/51190.
-
Pdgfra protects against ethanol-induced craniofacial defects in a zebrafish model of FASD. McCarthy N, Wetherill L, Lovely CB, Swartz ME, Foroud TM, Eberhart JK. Development. 2013 Aug;140(15):3254-65. doi: 10.1242/dev.094938.
-
Bmp and Shh signaling mediate the expression of satb2 in the pharyngeal arches. Sheehan-Rooney K, Swartz ME, Lovely CB, Dixon MJ, Eberhart JK. PLoS One. 2013;8(3):e59533. doi: 10.1371/journal.pone.0059533.